

Elementary Concepts and Fundamental Laws of the Theory of Heat
Types Of Pressure In Thermodynamics
History of Refrigeration and Refrigerators
History and Basics of Refrigeration
Heat Transfer for the Mechanical HVAC & Refrigeration PE Exam
HEATING AND AIR-CONDITIONING PRINCIPLES

The Boiling Point of Water can be changed and controlled by the controlling the vapor pressure above the water
Atmospheric Pressure: Pressure of the atmosphere at the earth’s surface NIST standard atmospheric pressure = 1.01325 bar.
Absolute Temperature: also called thermodynamic temperature, The temperature of an object on a scale where 0 is taken as absolute zero. Measured using the Kelvin Scale.
Kelvin Scale: an absolute thermodynamic temperature scale, meaning it uses absolute zero as its zero point
Absolute temperature and the kelvin scale
Fahrenheit: scale based on 32° for the freezing point of water and 212° for the boiling point of water, the interval between the two being divided into 180 equal parts
Celsius: also called centigrade, scale based on 0° for the freezing point of water and 100° for the boiling point of water
Rankine Scale:
British Thermal Unit (BTU): The Unit of Measurement for heat. the quantity of heat required to raise the temperature, of one pound of water, one degree Fahrenheit.
Conduction: Heat is transferred by direct contact
Convection: The transfer of heat through a medium like vapor or liquid
Radiation: Heats by waves or rays
Sensible Heat: The heat energy gained or released that causes a change in temperature and which can be measured
States of Matter- There are only three states or forms of matter: solid, liquid, and vapor/Gas.
Latent Heat: The heat energy required or released that causes a change of state while there is no change in temperature. It can not be measured with a thermometer.
Specific Heat: the heat required to raise the temperature of the unit mass of a given substance by a given amount (usually one degree).
Specific heat, heat of vaporization, and density of water
Saturation Temperature- The temperature of a liquid or vapor where if any heat is added or removed, a change of state will take place. Both liquid and vapor are present.
Boyle's Law - As the volume of refrigerant decreases the pressure increases. As the volume of refrigeration increases the pressure decreases. the pressure and volume of a gas are inversely proportional to each other as long as the temperature and the quantity of gas are kept constant.
Charles’ Law - As the pressure of refrigerant increases, so does the temperature. As pressure decreases in refrigerant, so does the temperature.
The ideal gas law (PV = nRT) relates the macroscopic properties of ideal gases. An ideal gas is a gas in which the particles do not attract or repel one another and take up no space (have no volume).
| = | pressure | |
| = | volume | |
| = | amount of substance | |
| = | ideal gas constant | |
| = | temperature |
Dalton’s Law - states that in a mixture of non-reacting gases, the total pressure exerted is equal to the sum of the partial pressures of the individual gases. If you combine two different gases, ie. refrigerant and nitrogen, their individual pressures add to each other, increasing pressure.
Law of Conservation of energy: Energy is neither created nor destroyed but can be converted from one form to the next
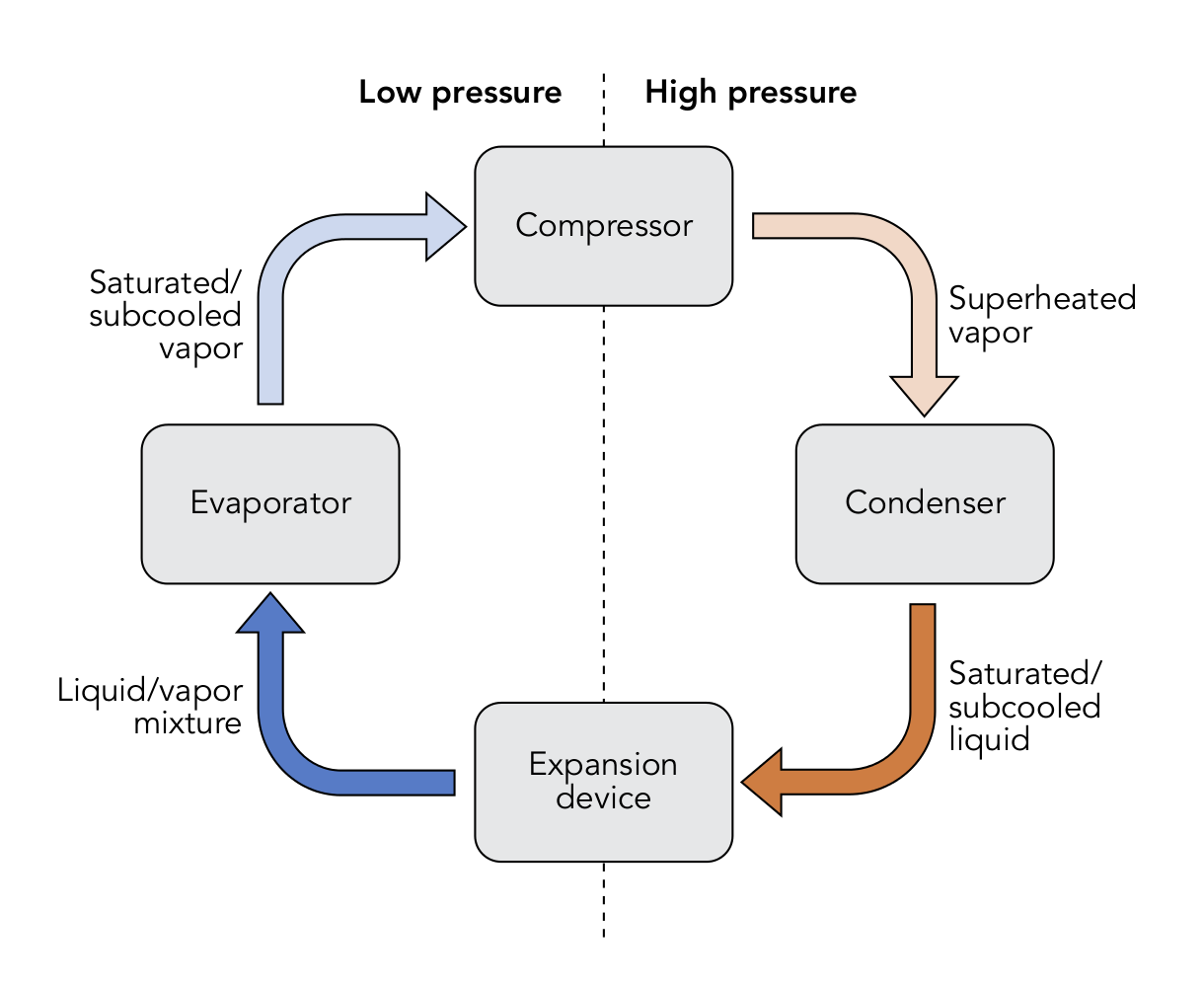
A refrigeration cycle has four major components:
A Refrigerant is a chemical compound capable of transitioning from liquid to gas and back again.